Diferencia entre revisiones de «Clorito de sodio»
m Revertidos los cambios de 189.143.223.113 (disc.) a la última edición de LMLM |
|||
| Línea 34: | Línea 34: | ||
==Fabricación== |
==Fabricación== |
||
The free acid, [[chlorous acid]], HClO<sub>2</sub>, is only stable at low concentrations. Since it cannot be concentrated, it is not a commercial product. However, the corresponding sodium salt, sodium chlorite, NaClO<sub>2</sub> is stable and inexpensive enough to be commercially available. The corresponding salts of heavy metals (Ag<sup>+</sup>, Hg<sup>+</sup>, Tl<sup>+</sup>, Pb<sup>2+</sup>, and also Cu<sup>2+</sup> and NH<sub>4</sub><sup>+</sup>) decompose explosively with heat or shock. |
|||
Sodium chlorite is derived indirectly from [[sodium chlorate]], NaClO<sub>3</sub>. First, the explosively (ONLY at concentrations greater than 10% in atmosphere) [[chlorine dioxide]], ClO<sub>2</sub> is produced by reducing sodium chlorate in a strong acid solution with a suitable reducing agent (for example, sodium chloride, sulfur dioxide, or hydrochloric acid). The [[chlorine dioxide]] is then absorbed into an alkaline solution and reduced with [[hydrogen peroxide]], H<sub>2</sub>O<sub>2</sub> yielding sodium chlorite. |
|||
==Uso== |
==Uso== |
||
The main application of sodium chlorite is the generation of chlorine dioxide for bleaching and stripping of textiles, pulp, and paper. It is also used for disinfection of a few municipal water treatment plants after conversion to [[chlorine dioxide]]. An advantage in this application, as compared to the more commonly used chlorine, is that [[trihalomethane]]s (such as [[chloroform]]) are not produced from organic contaminants. Sodium chlorite, NaClO<sub>2</sub> also finds application as a component in therapeutic rinses, mouthwashes, toothpastes and gels, mouth sprays, chewing gums and lozenges, and also in contact lens cleaning solution under the trade name purite. Under the brand name Oxine it is used for sanitizing air ducts and HVAC/R systems and animal containment areas (walls, floors, and other surfaces). |
The main application of sodium chlorite is the generation of chlorine dioxide for bleaching and stripping of textiles, pulp, and paper. It is also used for disinfection of a few municipal water treatment plants after conversion to [[chlorine dioxide]]. An advantage in this application, as compared to the more commonly used chlorine, is that [[trihalomethane]]s (such as [[chloroform]]) are not produced from organic contaminants. Sodium chlorite, NaClO<sub>2</sub> also finds application as a component in therapeutic rinses, mouthwashes, toothpastes and gels, mouth sprays, chewing gums and lozenges, and also in contact lens cleaning solution under the trade name purite. Under the brand name Oxine it is used for sanitizing air ducts and HVAC/R systems and animal containment areas (walls, floors, and other surfaces). |
||
| ⚫ | Sodium chlorite is also promoted as a miracle remedy to fight many diseases. After mixing a 28% sodium chlorite solution with a [[citric acid]] solution (activator) one becomes a "[[Miracle Mineral Supplement]]".<ref>{{cite news|url=http://www.brisbanetimes.com.au/queensland/backyard-cancer-healer-deceived-patients-20090423-agcy.html|title=Backyard cancer 'healer' deceived patients|last=Calligeros|first=Marissa|date=24 April 2009|publisher=The Brisbine Times|accessdate=28 December 2009}}</ref> |
||
El clorito de sodio también es promovido como un milagroso para combatir muchas enfermedades. Después de la mezcla de un 28% de solución de clorito de sodio con una solución de [[ácido cítrico]] (activador) uno se |
|||
| ⚫ | |||
En [[síntesis orgánica]], el clorito de sodio se utiliza frecuentemente para la oxidación de [[aldehído]]s a [[ácido carboxílico|ácidos carboxílicos]]. La reacción se realiza generalmante en un buffer (con [[fosfato monosódico]]) en la solución de la presencia de un captador de cloro (generalmente [[Penteno|2-metil-2-buteno]]). |
En [[síntesis orgánica]], el clorito de sodio se utiliza frecuentemente para la oxidación de [[aldehído]]s a [[ácido carboxílico|ácidos carboxílicos]]. La reacción se realiza generalmante en un buffer (con [[fosfato monosódico]]) en la solución de la presencia de un captador de cloro (generalmente [[Penteno|2-metil-2-buteno]]). |
||
Revisión del 23:46 18 ene 2010
| Clorito de sodio | ||
|---|---|---|
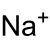 | ||
 | ||
| Nombre IUPAC | ||
| Clorito sódico | ||
| General | ||
| Fórmula semidesarrollada | NaClO2 | |
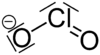
| Fórmula estructural |
| |
| Fórmula molecular | ? | |
| Identificadores | ||
| Número CAS | 7758-19-2[1] | |
| Número RTECS | VZ4800000 | |
| ChEBI | 78667 | |
| ChEMBL | CHEMBL1887585 | |
| ChemSpider | 22860 | |
| DrugBank | 13210 | |
| PubChem | 23668197 | |
| UNII | G538EBV4VF | |
| KEGG | C19523 | |
| Propiedades físicas | ||
| Apariencia | Sólido blanco | |
| Densidad | 2.5 g/cm3, solid kg/m³; Expresión errónea: palabra «g» desconocida g/cm³ | |
| Masa molar | 90.44 g/mol g/mol | |
| Punto de fusión | 180–200 °C decomp.' | |
| Propiedades químicas | ||
| Solubilidad en agua | 39 g/100 ml (17 °C) | |
| Valores en el SI y en condiciones estándar (25 ℃ y 1 atm), salvo que se indique lo contrario. | ||
El clorito de sodio es un compuesto químico utilizado en la fabricación de papel.
Fabricación
The free acid, chlorous acid, HClO2, is only stable at low concentrations. Since it cannot be concentrated, it is not a commercial product. However, the corresponding sodium salt, sodium chlorite, NaClO2 is stable and inexpensive enough to be commercially available. The corresponding salts of heavy metals (Ag+, Hg+, Tl+, Pb2+, and also Cu2+ and NH4+) decompose explosively with heat or shock.
Sodium chlorite is derived indirectly from sodium chlorate, NaClO3. First, the explosively (ONLY at concentrations greater than 10% in atmosphere) chlorine dioxide, ClO2 is produced by reducing sodium chlorate in a strong acid solution with a suitable reducing agent (for example, sodium chloride, sulfur dioxide, or hydrochloric acid). The chlorine dioxide is then absorbed into an alkaline solution and reduced with hydrogen peroxide, H2O2 yielding sodium chlorite.
Uso
The main application of sodium chlorite is the generation of chlorine dioxide for bleaching and stripping of textiles, pulp, and paper. It is also used for disinfection of a few municipal water treatment plants after conversion to chlorine dioxide. An advantage in this application, as compared to the more commonly used chlorine, is that trihalomethanes (such as chloroform) are not produced from organic contaminants. Sodium chlorite, NaClO2 also finds application as a component in therapeutic rinses, mouthwashes, toothpastes and gels, mouth sprays, chewing gums and lozenges, and also in contact lens cleaning solution under the trade name purite. Under the brand name Oxine it is used for sanitizing air ducts and HVAC/R systems and animal containment areas (walls, floors, and other surfaces).
Sodium chlorite is also promoted as a miracle remedy to fight many diseases. After mixing a 28% sodium chlorite solution with a citric acid solution (activator) one becomes a "Miracle Mineral Supplement".[2]
En síntesis orgánica, el clorito de sodio se utiliza frecuentemente para la oxidación de aldehídos a ácidos carboxílicos. La reacción se realiza generalmante en un buffer (con fosfato monosódico) en la solución de la presencia de un captador de cloro (generalmente 2-metil-2-buteno).
Recientemente, el clorito de sodio ha sido utilizado como un agente oxidante para convertir los furanos alquilo correspondientes a los 4-oxo-2-ácidos alquenoico en una síntesis simple una olla.[3]
Seguridad
El clorito de sodio, al igual que muchos agentes oxidantes, deben ser protegidos de la contaminación accidental de materiales orgánicos para evitar la formación de una mezcla explosiva.
Toxicidad
El clorito de sodio es un fuerte oxidante y por lo tanto, puede esperar que causen los síntomas clínicos similares a los del clorato de sodio, conocido: metahemoglobinemia, hemólisis, insuficiencia renal[4]. Una dosis de 10-15 gramos de clorato de sodio puede ser letal.[5]. La metahemoglobemia se había demostrado en ratas y gatos [6] y estudios recientes por la EMEA han confirmado que la sintomatología clínica es muy similar a la causada por el clorato de sodio en la rata, el ratón, el conejo y el Mono verde [7]
Solo hay un caso humano en la literatura médica de la intoxicación por clorito.[8]. Que parece confirmar que la toxicidad es igual al clorato de sodio. De la analogía con el clorato de sodio, incluso en pequeñas cantidades de alrededor 1 gramo puede esperarse a causar náuseas, vómito e incluso en peligro la vida de hemólisis en deficiencia de Glucosa-6-Fosfato Deshidrogenasa.
Referencias generales
- "Química de los Elementos", N.N. Greenwood y A. Earnshaw, Pergamon Press, 1984.
- "Kirk-Othmer Concise Encyclopedia of Chemistry", Martin Grayson, Editor, John Wiley & Sons, Inc., 1985
Referencias
- ↑ Número CAS
- ↑ Calligeros, Marissa (24 April 2009). «Backyard cancer 'healer' deceived patients». The Brisbine Times. Consultado el 28 December 2009.
- ↑ Annangudi SP, Sun M, Salomon RG (2005). «An efficient synthesis of 4-oxo-2-alkenoic acids from 2-alkyl furans» (abstract). Synlett 9: 1468. doi:10.1055/s-2005-869833.
- ↑ Goldfrank's Toxicologic Emergencies, McGraw-Hill Professional; 8th edition (March 28, 2006), ISBN 978-0071437639
- ↑ http://www.poisoncentre.be/article.php?id_article=39
- ↑ Clinical Toxicology of Commercial Products. Robert E. Gosselin, Roger P. Smith, Harold C. Hodge, Jeannet Braddock. Uitgever: Williams & Wilkins; 5 edition (September 1984) ISBN-13: 978-0683036329
- ↑ Sodium Chlorite - Summary Report of the Europena Agency for the Evaluation of Medicinal Products - Veterinary Medicines Evaluation Unit [[1]]
- ↑ Acute sodium chlorite poisoning associated with renal failure. Lin JL, Lim PS. Ren Fail. 1993;15(5):645-8. PMID: 8290712
- Esta obra contiene una traducción derivada de «Sodium chlorite» de Wikipedia en inglés, publicada por sus editores bajo la Licencia de documentación libre de GNU y la Licencia Creative Commons Atribución-CompartirIgual 4.0 Internacional.
